GeneOne Life Science, which hit the daily upper limit, drew attention after disclosing it had secured won 6.6 billion in operating funds. Y2Solution saw heightened investor sentiment on expectations for first-quarter revenue. Voronoi gained attention after presenting positive Phase 1 results for its lung cancer treatment candidate “VRN11” at the American Association for Cancer Research (AACR).
|
◇GeneOne Life Science secures won 6.6 billion via CBs
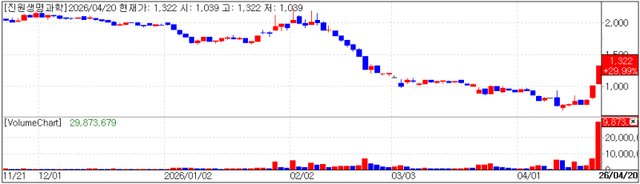
KOSPI-listed GeneOne Life Science closed up 29.99% (won 305) at won 1,322, with a market capitalization of won 120.1 billion.
The company has been posting operating losses on both separate and consolidated bases, resulting in negative cash flow. As of end-2024, its cash holdings stood at just won 1 billion, raising liquidity concerns.
On April 20, the company announced it would secure a total of won 6.6 billion in cash through the issuance of new convertible bonds (CBs) and the sale of treasury CBs. This is expected to cover near-term operating expenses. However, as CBs are classified as liabilities, they may increase the company’s financial burden.
Specifically, the company will sell won 1.5 billion worth of treasury CBs to Seonkwang System and issue new CBs worth won 2.1 billion to SW Entertainment and won 3 billion to Dooyoung Mulsan.
GeneOne had repurchased won 3 billion worth of its 6th CB issued in November last year before maturity, and decided to resell won 1.5 billion of it to Seonkwang System. The conversion price for the CB purchased by Seonkwang System is set at won 2,192, while newly issued CBs to SW Entertainment and Dooyoung Mulsan are set at won 1,065.
Meanwhile, GeneOne Life Science received penalty points for disclosure violations after delaying the announcement of a change in its largest shareholder. The latest penalty of 4 points adds to a previous 5 points, bringing the cumulative total to 9. If the total exceeds 15 points within a year, the company may be designated as a managed stock.
The largest shareholder changed on Feb. 13 but was disclosed late on March 17. Former largest shareholder Park Young geun, head of the Life Science division, sold about 1.16 million shares (worth approximately won 2.2 billion), reducing his stake from 6.23% to 4.87%. As a result, Dongban Growth Investment Association No.1, previously the second-largest shareholder with a 5.73% stake, became the largest shareholder.
Some market participants classify GeneOne as a COVID-19 beneficiary stock. The company develops DNA vaccine platforms and gene-based vaccines. With signs of the spread of the BA.3.2 COVID-19 variant, also known as “Cicada,” vaccine developers like GeneOne are being revisited.
According to the Korea Disease Control and Prevention Agency, BA.3.2 is a subvariant of Omicron first identified in South Africa in November 2024. Its detection rate in Korea rose from 3.3% in January to 12.2% in February and 23.1% in March. Authorities say it is not yet a major concern.
|
◇Y2Solution buoyed by Q1 revenue expectations
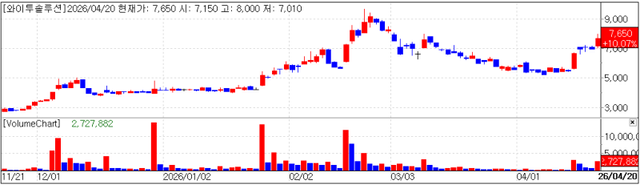
Y2Solution rose 10.07% (won 700) to close at won 7,650. It had climbed about 15% intraday before trimming gains. The stock appears to have drawn attention as a PharmEdaily pay-to-read article published earlier on April 12 titled “Y2Solution’s Q1 revenue outlook…‘quantum jump’ becoming reality?” was made available on portal sites.
According to the article, Y2Solution is expected to post strong first-quarter revenue, with some estimates suggesting it could achieve more than half of last year’s annual revenue of approximately won 160 billion. If realized, this would demonstrate that the company’s target of doubling revenue this year is not merely aspirational.
Last year, Y2Solution recorded revenue of won 165 billion, up 6.8% year over year. However, operating profit swung to a loss of won 2.5 billion from a profit of won 2.8 billion, and net profit also turned to a loss of won 9.5 billion from a profit of won 6.5 billion.
The company attributed the decline to weak performance among clients in its core power supply unit (PSU) business and intensified price competition among vendors, which hurt profitability. It said aggressive cost-cutting efforts helped minimize losses.
Expectations for Q1 performance are driven by its biofuel business. After entering the market in the second half of last year, the segment generated over won 40 billion in revenue within less than six months and is continuing rapid growth this year.
Y2Solution is currently active in the distribution of biodiesel feedstock, including soybean oil, corn, used cooking oil, and animal fats. Its logistics system, built through partnerships with major domestic petrochemical manufacturers, is creating strong synergies in the biofuel market.
Attention is also on its U.S. subsidiary Luxa Biotechnology’s dry age-related macular degeneration treatment, RPESC-RPE-4W, currently in Phase 1/2a trials. The therapy uses allogeneic retinal pigment epithelium (RPE) stem cells and is being tested in 18 subjects.
Y2Solution invested $6.5 million (about won 10 billion) in Luxa through a joint investment agreement with the U.S. based nonprofit Neural Stem Cell Institute in 2019 and holds a 50% stake.
Dukwoo Electronics is the largest shareholder with a 28.44% stake, followed by The Best Resource (8.03%), JPMorgan Securities (1.18%), and Merrill Lynch International (1.01%).
|
◇Voronoi rises on AACR updates for VRN10, VRN11
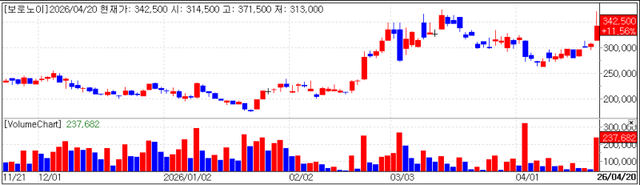
Voronoi rose 11.56% (won 35,500) to close at won 342,500. Investor sentiment strengthened after the company announced positive data for its key pipelines VRN10 and VRN11 at AACR 2026, held in San Diego from April 17 to 22.
According to Voronoi, VRN11, a next-generation targeted therapy for lung cancer, showed a 100% objective response rate (ORR) in patients with EGFR C797S resistance. All six patients receiving doses of 160 mg or higher achieved tumor reduction (partial response). Disease control was observed in 96.8% of EGFR-mutant patients (30 out of 31).
VRN11 demonstrated meaningful Phase 1 results in patients with EGFR C797S mutations, a common resistance mechanism following treatment with third-generation EGFR inhibitor Tagrisso (osimertinib). The company believes VRN11 has entered the visible range of becoming a first-line treatment for lung cancer, with improved efficacy and safety over existing therapies.
Safety data were also encouraging. Among 65 patients receiving doses ranging from 10 mg to 480 mg, only one case of drug-related grade 3 or higher adverse events was reported. Notably, no grade 3 or higher adverse events occurred in patients receiving doses of 320 mg or higher. Data from patients treated for nearly one year also support its long-term safety profile.
Based on these results, Voronoi plans to initiate a Phase 1b/2 trial cohort for EGFR C797S mutation patients within the year, aiming for Accelerated Approval based on Phase 2 data, given the lack of approved treatments.
For VRN10, the company highlighted new potential in breast cancer treatment. VRN10 was found to induce HER2 degradation, differentiating it from existing therapies. This mechanism enhances potential synergy with antibody-drug conjugates (ADCs) such as Enhertu.
In combination with Enhertu, VRN10 promotes lysosomal degradation of the HER2-Enhertu complex, increasing the release of the cytotoxic payload (DXd) and enhancing cancer cell death. This mechanism is considered a key differentiator compared to competitors such as Tukysa, Hernexeos, and Hyrnuo.
Voronoi is currently conducting dose escalation up to 480 mg for VRN10, with the data expected to accelerate future clinical development. The company plans to launch Phase 1b/2 trials this year to optimize dosing for both monotherapy and combination with Enhertu.
Copyright ⓒ 이데일리 무단 전재 및 재배포 금지
본 콘텐츠는 뉴스픽 파트너스에서 공유된 콘텐츠입니다.