|
◇PeopleBio shares hit the upper limit after receiving an unqualified audit opinion
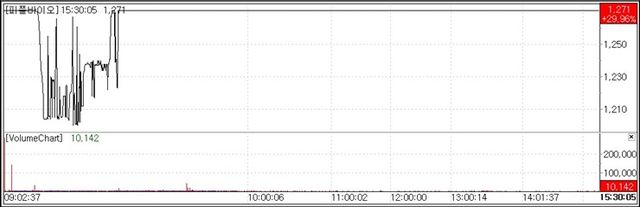
According to KG Zeroin’s MP Doctor on April 9, shares of Alzheimer’s blood diagnostic firm PeopleBio rose 29.96%(293 won) to close at 1,271 won. The rally was driven by the company securing an unqualified audit opinion.
The company disclosed that it received an unqualified opinion from its external auditor for its 2025 fiscal year audit report. It was also removed from the investment caution designation effectively eliminating one of the biggest overhangs financial and accounting risks. Following the announcement the stock hit the daily price limit.
PeopleBio stated that it had focused on restructuring its business and enhancing transparency in accounting. By applying conservative accounting standards and proactively reflecting potential financial risks the company said it resolved uncertainties surrounding its financial condition.
The market interpreted the audit opinion upgrade and removal from the caution list as a “risk-off event.” In the biotech sector, resolving accounting issues often leads to a release of investment restrictions and a surge in short term liquidity, which appears to have driven the stock’s sharp rise.
However industry observers noted that an unqualified audit opinion merely confirms that financial statements are properly prepared and does not validate business performance or growth potential. Going forward commercialization of PeopleBio’s Alzheimer’s blood diagnostic technology and revenue expansion will be key determinants of its valuation.
The company plans to focus on improving profitability and stabilizing operations centered on its core business. It also aims to create synergies by integrating its Alzheimer’s diagnostic platform with IT and AI businesses, including an edge data center project in Samsung dong led by its major shareholder RealityZen.
A company official said “Receiving an unqualified audit opinion and being removed from the caution list mark the beginning of normalization. We will focus on delivering tangible results.”
◇Megagen Acquires 5% Stake in Ray, Citing ‘Management Influence’ Shares Hit Limit-Up
Ray also drew strong investor attention after Megagen Implant acquired a stake in the company. On the same day Ray shares surged 29.93% to close at 6,490 won hitting the upper limit. The move was driven by expectations of strategic investment.
Megagen disclosed on April 8 via the Financial Supervisory Service’s DART system that it had acquired shares in Ray, stating the purpose as “influence on management.” The stake accumulation was not a one-time purchase but had been ongoing since January this year.
Industry sources highlighted that Megagen acquired the shares without prior notice or consultation with Ray. Given that strategic investments are typically preceded by coordination, this move is being interpreted as potentially opening the door to active management participation rather than a purely financial investment.
Ray is recognized for its technological competitiveness in digital dental imaging equipment, including cone beam computed tomography(CBCT). Analysts suggest that Megagen’s investment reflects a strategic move to expand beyond implants into diagnostic equipment. Market participants are also discussing potential synergies such as shared sales channels and bundled offerings.
The market views Megagen’s stake acquisition as more than a simple investment, pointing to the potential integration of diagnostics and treatment within the dental value chain. Future collaboration and realization of business synergies are key factors investors will be watching.
◇GemVax Falls Despite Phase 2a Extension Study Results
GemVax declined despite announcing results from its extension study for GV1001, a treatment candidate for progressive supranuclear palsy(PSP). The stock fell 7.49%(2,250 won) to close at 27,800 won.
The company announced that it had received the Clinical Study Report(CSR) for its domestic Phase 2 extension trial. According to GemVax, the CSR confirmed not only safety but also statistically significant results in key PSP symptoms, including cognition, ocular motor function, and limb motor function.
However, the market reaction was negative. While the study met safety endpoints including adverse events, laboratory tests, vital signs and ECG detailed efficacy data were not fully disclosed, which appears to have weighed on investor sentiment.
The 72 week study consisted of a 24-week main trial followed by a 48 week extension. The company stated that meaningful outcomes were included in the CSR based on comparisons with external control groups and detailed analyses.
In patients with Richardson’s syndrome(PSP-RS), the low-dose group showed statistically significant improvement in cognition compared to the placebo group from the prior study. T-tests also indicated statistically significant differences(p-value < 0.05) in cognition, ocular motor function, and limb motor function versus placebo.
A T-test is a statistical method used to determine whether differences between two groups are meaningful rather than due to chance. A p-value below 0.05 is generally considered statistically significant.
A GemVax official said, “Given the positive CSR results, we expect a smoother regulatory approval process with global authorities such as the FDA. We also anticipate this will accelerate investment and partnerships with global companies.”
Copyright ⓒ 이데일리 무단 전재 및 재배포 금지
본 콘텐츠는 뉴스픽 파트너스에서 공유된 콘텐츠입니다.