Samchundang Pharmaceutical fell approximately 5% despite CEO Jeon In-seok holding an emergency press conference and announcing the cancellation of a 250 billion won block deal. Rokit Healthcare plummeted 23%, while Caregen dropped 16%. The lingering skepticism surrounding Samchundang once the top cap stock on the KOSDAQ appears to be casting a shadow over broader biotech sentiment.
|
◇Samchundang Pharma, CEO Jeon In-seok Says "S-PASS Is Real"
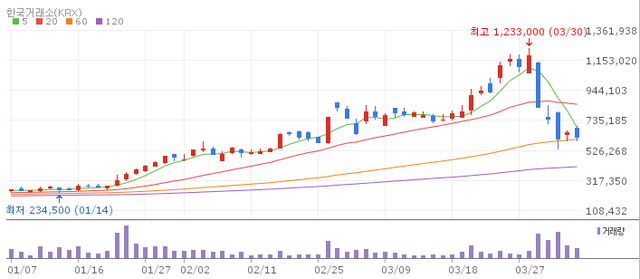
According to KG Zeroin MP DOCTOR data Samchundang Pharmaceutical's share price fell roughly 5% on the day, trading at 618,000 won in after-hours markets. At its peak in late March the stock had soared to over 1.23 million won, briefly claiming the top market-cap spot on the KOSDAQ meaning the stock has lost roughly half its value in under two weeks.
CEO Jeon In seok convened an emergency press briefing at the company's headquarters in Seocho-gu, Seoul, directly addressing the controversy over his share sale plan, the authenticity of the company's proprietary S-PASS drug delivery technology, and concerns about the reliability of the company's disclosures.
He opened by announcing the complete withdrawal of the 250 billion won block deal that the largest shareholder had planned to execute.
"Proceeding with the sale while the stock has fallen more than 30% would only further erode shareholder trust" he said while reiterating that the original plan was solely intended to cover inheritance and gift tax obligations, and was entirely unrelated to any change in management control or the company's fundamentals.
On the most contentious issue the legitimacy of the S-PASS platform Jeon produced official documents submitted to the U.S. Food and Drug Administration (FDA), declaring flatly, "S-PASS is not a fake technology". "The documents submitted to the FDA contain a detailed explanation of how our technology works.
If this were fraudulent the FDA would never have accepted the filing" he said adding "What we have is not a mere concept it is technology that has already been reviewed by a global regulatory authority."
The disclosed documents reference the S-PASS patent number, an Abbreviated New Drug Application (ANDA) filing, and the notation "SNAC-Free" which the company presented as evidence of its independent technological differentiation.
Addressing criticism over what many investors perceived as premature or unverified public disclosures, Jeon offered a formal apology. "I believed that if we focused on our products and technology, the sincerity of our work would speak for itself.
In doing so I neglected to communicate adequately with shareholders and the market and that was my mistake" he said bowing in contrition. He also pledged a full overhaul of the company's disclosure and investor relations processes including quarterly IR sessions, mandatory preconsultation with the Korea Exchange (KRX) before announcing material contracts and the creation of a dedicated PR/IR team.
Jeon also clarified the structure of the oral semaglutide supply agreements that had drawn scrutiny. "These contracts are not simple technology transfers. We supply the active ingredients and proprietary technology while the partner handles commercialization and if the partner's sales fall below 50% of the agreed target Samchundang retains the right to terminate the contract" he explained emphasizing that the company holds structural leverage.
He further noted that partners in the U.S., Europe and Japan each conducted independent legal and patent due diligence to verify the company's ability to design around the originator's patents before signing.
On the road ahead, Jeon stated the company plans to file an Investigational New Drug (IND) application next month expects to receive final clinical study results between late Q3 and early Q4 of this year, and aims to secure at least two additional global supply agreements before year end.
However market participants remain cautious with key uncertainties including the KRX's ongoing review of the company's disclosure conduct and the outcome of pending legal disputes.
|
◇Rokit Healthcare, Post Tenbagger Correction Hits Hard
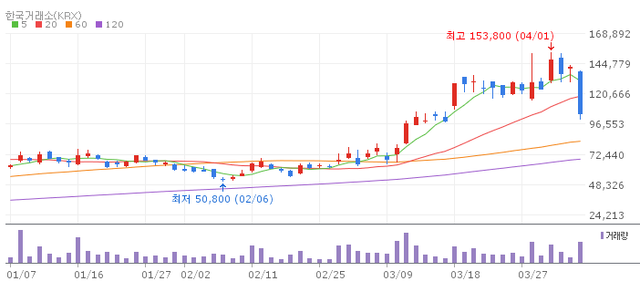
Rokit Healthcare tumbled approximately 23% on the day, settling at 109,300won in after hours trading.
Having surged more than 10-fold from its IPO price of 11,000 won within less than a year of its KOSDAQ listing in May 2025, the stock's steep decline is widely attributed to a wave of profit taking following the rapid run up, compounded by broader market volatility.
Circuit breakers (Volatility Interruption, or VI) have been triggered repeatedly in recent sessions reflecting the extreme swings in trading activity.
Rokit Healthcare develops tissue and organ regeneration platforms by integrating 3D bioprinting with artificial intelligence, with applications spanning knee cartilage, skin, and pancreatic islet regeneration.
The company achieved its first operating profit in 2025 approximately 600 million won but recorded a net loss due to convertible bond valuation losses, a point of ongoing concern for investors monitoring the balance sheet.
IBK Investment & Securities has maintained a constructive long term view stating that "the growth thesis underpinned by the regenerative medicine platform remains valid" while cautioning that "a conservative approach is warranted during this high volatility phase following the sharp run up."
Over the medium to long term progress on domestic and international clinical trials, advancement in global patent and regulatory approvals, and the expansion of overseas partnerships are seen as the key variables that will determine the stock's direction.
Market views are split between those who expect the growth narrative to reassert itself after the technical correction and those who remain concerned about stretched valuations relative to current fundamentals.
|
◇Caregen, Earnings Miss and Short Selling Pressure Drive 16% Decline
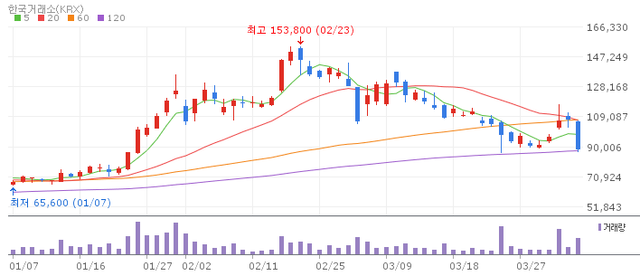
Caregen declined 16% to close at approximately 90,000won. The synthetic peptide-based biocosmetics and pharmaceutical ingredient company has been under sustained selling pressure amid a combination of weak earnings, short selling activity and regulatory designations.
In Q4 2025 (consolidated basis), the company reported revenue of 16.3 billion won a 24% year on year decline alongside an operating loss of 7.1 billion won and a net loss of 3.3 billion won badly missing market expectations.
In March the company filed a disclosure noting that its revenue or profit structure had changed by more than 30%, and the repeated designation and re designation of the stock as a "short-selling overheated issue" has further dampened sentiment. Some analysts suggest the correction reflects a broader revaluation of the company's premium multiple in the context of deteriorating profitability.
On the positive side, management presented a medium-term shareholder value enhancement plan at the annual general meeting, committing to annual operating profit growth of at least 5%, a dividend payout ratio of 30% or more, and a sustained return on equity (ROE) of 15% or above.
One biotech analyst, speaking on condition of anonymity, commented: "Progress on the clinical and regulatory front for pipeline candidates such as CG-P5, along with a recovery in overseas sales particularly in the U.S. and China will be the key catalysts to watch for a meaningful stock recovery.
The company's proprietary peptide technology and global network give it room to reclaim a valuation premium if fundamentals recover but the short selling and regulatory overhang means a cautious approach is still warranted for now".
Copyright ⓒ 이데일리 무단 전재 및 재배포 금지
본 콘텐츠는 뉴스픽 파트너스에서 공유된 콘텐츠입니다.