|
◇ImmuneOncia Records 63% Complete Response Rate in Denverstotug Phase 2 Trials
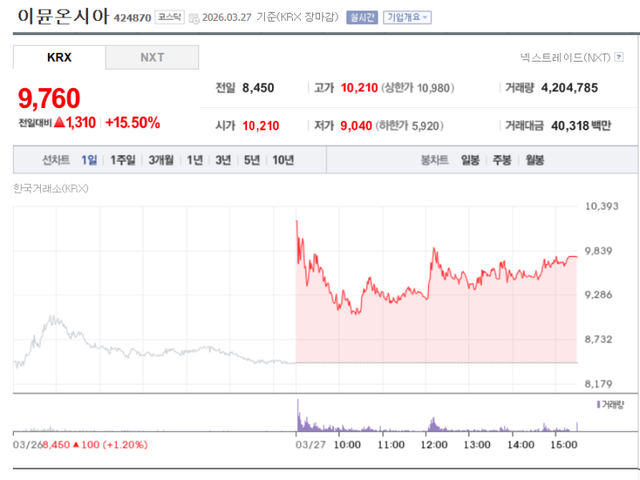
According to KG Zeroin MP DOCTOR, ImmuneOncia closed at 9,760 won, up 15.50% from the previous day. The main catalyst for this surge was the Phase 2 clinical results of its immuneoncology candidate, Denverstotug (MC-001), presented as a poster at the Korean Society of Hematology International Conference (KSHIC 2026).
According to the data disclosed by ImmuneOncia, Denverstotug monotherapy for patients with relapsed or refractory NK/T-cell lymphoma a rare and incurable blood cancer with no standard treatment recorded an objective response rate (ORR) of 79% and a complete response rate (CR) of 63%, meaning total tumor disappearance.
While NK/T-cell lymphoma is considered a disease with a poor prognosis with existing chemotherapy offering a median progression-free survival (mPFS) of only 4.1 months Denverstotug demonstrated exceptional therapeutic efficacy in this trial recording an mPFS of 29.4 months and a median overall survival (mOS) of 40.2 months. Notably a 2year survival rate of 78% was observed, and 40% of all patients completed two years of treatment without significant adverse events, underscoring the drug’s superior safety profile.
A standout feature of this presentation was the analysis results of the tumor microenvironment (TME) and Membrane Specificity (MS) using Artificial Intelligence (AI). Contrary to conventional wisdom, high therapeutic responses were observed even in patients with low PD-L1 expression.
This suggests that the MS index, which analyzes membrane patterns rather than simple expression levels, could serve as a new precision medicine biomarker. Medical experts, including Professors Won-seok Kim and Seok-jin Kim of the Department of Hematology Oncology at Samsung Medical Center evaluated these results as highly encouraging for a monotherapy in the absence of standard treatment and expressed hope for rapid commercialization to provide a desperate alternative for patients.
ImmuneOncia completed the orphan drug designation for Denverstotug with the Ministry of Food and Drug Safety (MFDS) last January and plans to accelerate the approval process by applying for Global Innovative Products Fast Track (GIFT) within this year.
Notably, the company proved its technological prowess and commercial value by signing a commercial development agreement through 2030 with Lonza, a global contract development and manufacturing organization (CDMO).
Heung tae Kim CEO of ImmuneOncia noted that the Phase 2 trial proved Denverstotug is an innovative treatment for blood cancer patients with no other options, and expressed his commitment to launching Korea’s first domestic immune-oncology drug and expanding into the global market within three years through close cooperation with Lonza.
|
◇Toolgen Enters "Main Event" of Patent Dispute in H2… Secures Senior Party Status
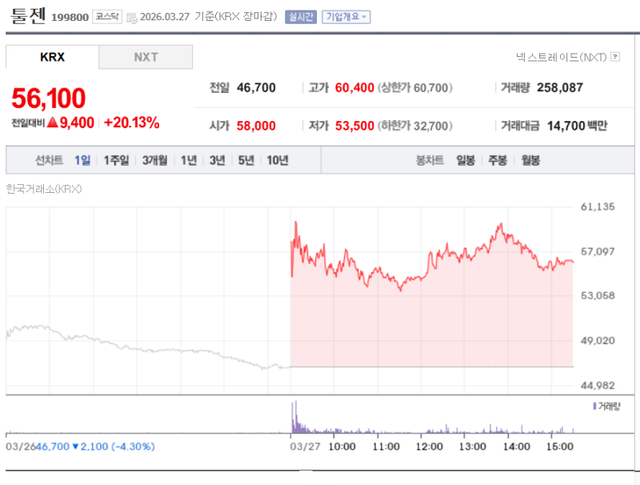
Toolgen also ended the day’s trading at 56,100 won, a 20.14% surge from the previous day. This was largely driven by recent interview remarks from Toolgen CEO Josang Yu, who suggested that the decisive turning point in the global CRISPR gene-editing patent dispute is approaching in early July or August.
Yu anticipated that the second phase of the interference proceeding (Priority Phase) the priority determination process of the U.S. Patent Trial and Appeal Board (PTAB) that had been delayed will soon resume. This is effectively the "main event" that determines who first implemented the technology in eukaryotic cells.
The current CRISPR-Cas9 eukaryotic cell original patent dispute is a decade-long global legal battle between Toolgen, the Broad Institute (MIT/Harvard), and the CVC Group (UC Berkeley). While industry attention has been high since the final winner will secure core rights in the global gene editing market, the litigation had been sluggish.
However the atmosphere shifted in January when the PTAB accepted Toolgen’s request to expedite the interference proceedings despite opposition from competitors. Previously the structure required the conclusion of the CVC-Broad interference before proceeding with Toolgen but this decision has reduced the risk of long-term delays.
Currently, Toolgen holds "Senior Party" status in the first stage of the interference proceeding. According to the company, this puts them in a structurally advantageous position as it means the opposing parties bear the initial burden of proof in the priority dispute.
It is also expected that while the main arguments and evidence of the opposing camps have been largely disclosed, Toolgen’s strategy remains relatively less exposed.
Industry analysts believe that entering the Priority Phase will significantly increase litigation costs and risks for all parties, which, ironically, may increase the incentive to reach a settlement. Since uncertainty in patent rights creates barriers to commercialization, stakeholders may eventually be forced to consider settlements that divide market shares.
In preparation, Toolgen has been shifting its strategic focus from patent prosecution to active rights enforcement, recently appointing litigation expert Bon-cheon Ku as Chief Legal Officer (CLO).
Furthermore, Toolgen is pursuing a "two-track strategy" utilizing its original patent alongside its RNP (ribonucleoprotein) patent, which offers high safety and accuracy. Based on this technology, which is already applied in commercialized gene editing therapies, the company has secured rights in the U.S. and is pursuing infringement litigation against related firms.
Both companies saw a strong influx of buying interest as they provided concrete evidence for growth proven clinical data and progress in international patent proceedings rather than mere vague expectations.
Copyright ⓒ 이데일리 무단 전재 및 재배포 금지
본 콘텐츠는 뉴스픽 파트너스에서 공유된 콘텐츠입니다.